DESULFURIZATION SYSTEM
Converting Lead Sulfate for Cleaner Smelting
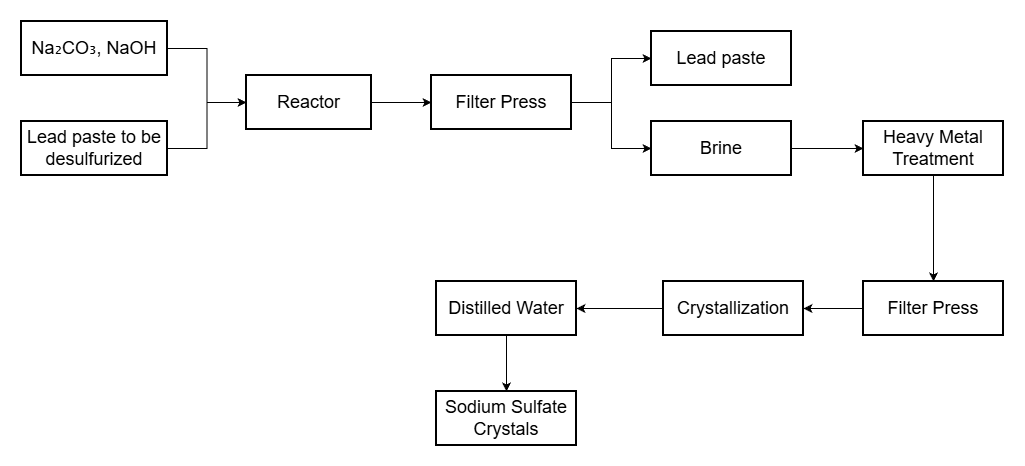
Lead paste from spent batteries contains significant lead sulfate (PbSO₄). Direct smelting generates large amounts of SO₂, requiring expensive flue gas treatment. Desulfurization converts lead sulfate to lead carbonate (PbCO₃) or lead oxide (PbO), enabling low-temperature smelting with minimal sulfur emissions.
Our desulfurization process uses sodium carbonate or sodium hydroxide solutions, achieving over 95% conversion rate. The byproduct sodium sulfate can be crystallized and sold, turning waste treatment into value creation.